Hello ADNP community, families and friends, we anticipate that 2024 will mark a pivotal year in drug discovery for ADNP, offering a multitude of potential treatments and significantly advancing our goals. We are excited at what the future holds for our children.
On Tuesday, November 28th, nonprofits, including ours, will unite to seek your support. While there is no shortage in worthwhile causes, we urge you to rally behind ADNP syndrome, as it critically requires your support for a cure.
Donations for ADNP syndrome research have hit their lowest point in years. As we approach the end of the year, we are determined to reverse this trend.
WHY DONATE? Below are exciting research project updates.
The most recent animal model characterization projects reveal promising developments. The Buxbaum US mouse line, featuring one of the syndrome's most prevalent mutations, shows a robust phenotype, including early tooth eruption—a significant biomarker for ADNP, which we are celebrating in the photo below.
Successes toward drug treatment discovery has yielded two new groups of promising candidates are undergoing preliminary validations, with confirmation expected in Q1 2024 from Mount Sinai. Additionally, out of Mount Sinai, an exciting gene specific therapy project targeting the most prevalent mutation in ADNP syndrome is in progress.
In October, contracts were finalized to fund the 2nd $150,000 phase 2 of the RareBase FUNCTION high throughput screening project. Phase 1 resulted in a very positive list of over 100 possible treatments for ADNP syndrome. The team has hand selected the top 25 and will begin on milestone one, which is concentration response confirmation testing on 26 drug candidates in wild-type ADNP neuronal cell models. From there, 3 lead candidates will be extensively tested, which we hope find one or more solid possible treatment options for ADNP.
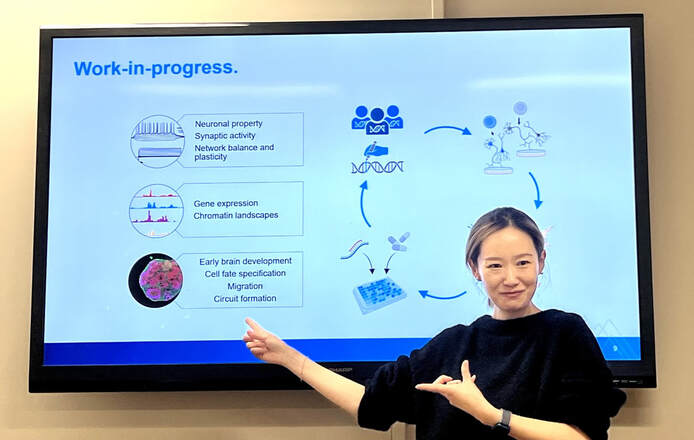
Molecular phenotyping, initiated at the beginning of the year, aims to identify electrophysiological changes in ADNP as potential biomarkers and outcome measures for treatment efficacy.
Molecular phenotyping, initiated at the beginning of the year, aims to identify electrophysiological changes in ADNP as potential biomarkers and outcome measures for treatment efficacy.
One of the most anticipated reports encompasses the Seaver team's ongoing work on the Low Dose Ketamine drug trial in ADNP. Dr. Kolevzon reported a discovered biomarker for the trial's disease-modifying potential. Although the team did not secure the first grant, a new application with refined outcome measures has received a favorable initial response, and final NIH results are anticipated in early 2024. The Seaver team is also exploring Organoids as a 3D model for studying and screening compound libraries in ADNP-deficient organoids, with promising results and plans to initiate screening by the year's end.
The UC Davis/MIND team is actively engaged in our gene therapy projects. While no updates are available currently, the team is testing Aims on ADNP neurons and is prepared to conduct mouse trials following the completion of characterization, with a comprehensive report scheduled for late January.
The cost of drug development is exorbitant. Basic research, drug discovery, and pre-clinical data for just one therapeutic candidate typically range from $500,000 to $2 million (data from FAST).
Every donation, regardless of size, propels our mission to cure ADNP syndrome, advocate for those affected and awareness, and enhance the quality of life for individuals living with ADNP. PLEASE GIVE NOW ON THIS GIVING TUESDAY!